Fractyl Health (GUTS) Surges 21% After Hours: Key Insights
Fractyl Health’s Stock Surges After Q4 Results and New Data
Fractyl Health Inc. (GUTS) experienced a significant surge in after-hours trading on Tuesday, with the stock rising 20.89% to $0.52. This increase followed the company’s release of fourth-quarter 2025 results and new data from its REMAIN-1 Midpoint Cohort, a pilot study evaluating an investigational outpatient, non-drug procedure designed to maintain weight loss following GLP-1 therapy.
Key Highlights from Q4 Results
For the fourth quarter of 2025, Fractyl reported several key metrics compared to the same period in the previous year. The company noted that research and development spending decreased as priorities shifted in the first quarter of 2025. However, selling, general, and administrative costs increased due to financing commissions in August 2025. The net loss for the quarter was primarily driven by a $20.2 million non-cash adjustment related to warrant liabilities.
Full-Year Performance Overview
Looking at the full year of 2025, Fractyl reported cash and equivalents of $81.5 million as of December 31, 2025. Additionally, the company received $4.1 million in subsequent warrant exercise proceeds in January 2026. These figures indicate that the combined cash position is expected to fund operations into early 2027, extending beyond the anticipated pivotal data readout.
Dose-Response Signal Strengthens Pivotal Confidence
New post-hoc analyses from the REMAIN-1 Midpoint Cohort revealed a statistically significant link between ablation length and weight maintenance in the Revita arm—the company’s investigational endoscopic duodenal remodeling procedure. In the highest-dose subgroup, participants using Revita maintained 88% of their previous weight loss after six months. This finding has bolstered confidence in the potential efficacy of the procedure.
FDA Pathway and Regulatory Progress
Fractyl reported that it received pre-submission feedback from the U.S. Food and Drug Administration (FDA). The agency acknowledged the safety profile of Revita, based on data from over 300 procedures, as consistent with a Class II device classification. Class II devices are moderate-risk medical devices that require both general controls and special controls to ensure safety and effectiveness.
The company plans to submit a potential De Novo marketing application for Revita in the late fourth quarter of 2026, pending the results of pivotal data.
Trading Metrics and Technical Analysis
As of the latest data, Fractyl Health has a market capitalization of $65.41 million. The stock’s 52-week high stands at $3.03, while its 52-week low is $0.38. The Relative Strength Index (RSI) for the Massachusetts-based metabolic therapeutics company is currently at 27.33. Over the past 12 months, the small-cap stock has declined by 65.01%, bringing it close to its annual low.
The ongoing decline and current positioning suggest that any potential recovery would need clear confirmation before investors consider stepping in.
Price Action and Market Trends
Fractyl closed the regular session 3.48% lower at $0.43, according to Jendela MagazinePro data. Jendela Magazine’s Edge Stock Rankings indicate that GUTS has a negative price trend across all time frames.
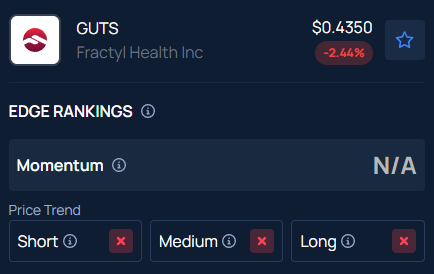
Image via Shutterstock/ ZCOOL HelloRF
Disclaimer
This content was partially produced with the help of AI tools and was reviewed and published by Jendela Magazine editors.
© 2026 Jendela Magazine. Jendela Magazine does not provide investment advice. All rights reserved.
Sponsored Content
Retirement can be a difficult part of life to navigate, and a financial advisor can help. Finding a qualified financial advisor doesn’t have to be hard. SmartAsset’s free tool matches you with up to three financial advisors who serve your area, and you can interview your advisor matches at no cost to decide which one is right for you. If you’re ready to find an advisor who can help you achieve your financial goals, get started now.